Introducing: ONI’s Next-Generation End-to-End Solution for EV Research Learn More
Biosafety Cabinets
Challenge
Enabling super-resolution microscopy imaging in biosafety cabinets
Super-resolution microscopy has provided great breakthroughs in multiple areas of biomedical research, thanks to its ability to enhance the resolution of conventional fluorescence microscopy. Due to their size and design the majority of super-resolution microscopes have not able to support research in enclosed biosafety cabinets. Ventilated biosafety cabinets are necessary when working with materials potentially contaminated with pathogens or under stringent sterile conditions (i.e. GMP labs).

Solution with the Nanoimager
The next generation microscope now operating in biosafety cabinets
The Nanoimager is the world’s first desktop super-resolution microscope. It is a class 1 laser-safe, compact instrument (A4 footprint) that can capture the most sensitive single-molecule fluorescence data even in a biosafety cabinet. The Nanoimager enables live-cell imaging over extended time periods, with optional whole body heating, Z-lock and microfluidic device support. Our NimOS software includes a tracking tool that allows users to visualize tracks, calculate diffusion coefficients, size particles and measure their concentration, both in live cells and in solution.
For the strictest environments, the Nanoimager allows researchers to limit contact time with infectious samples through its automation capability and remote control. Control of the sample position is highly intuitive within the user interface: sample exploration can be performed using the keyboard or an Xbox controller, requiring no direct wiring into the microscope. Additionally, it has an alignment design that does not require any re-adjustments over time.
Overall, the compact size, unrivalled stability and ability to work in so many different environments make our microscope a strong choice for BSL3 safety cabinets and BSL4 facilities.

Case Study
Imaging tubulin in HaCaT cells using dSTORM in a biosafety cabinet
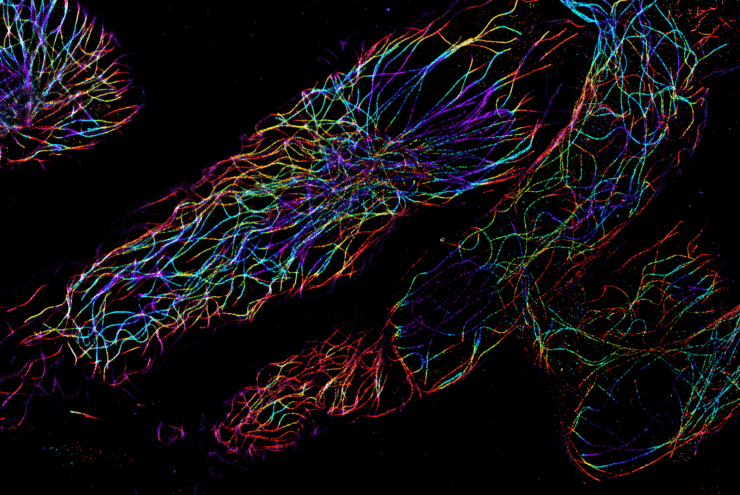
dSTORM imaging offers the opportunity to see inside the cell or look at purified viral particles with a resolution of up to 20 nm using localization-based super-resolution microscopy. With the Nanoimager, the spatial relationship of up to four molecular species can be imaged and quantified (with four separate laser lines) and imaging can be performed with two colors simultaneously.
In this figure we present dSTORM images of tubulin labelled with AF647 in HaCaT cell, acquired inside the safety cabinet (A) on a lab bench (B). In the right panels, individual localizations have been colored according to the frame of their appearance (1-20000 frames). The figure demonstrates an extremely low level of sample drift considering the acquisition of data over tens of thousands of frames.
Learn more about the Nanoimager’s single-particle tracking and the super-resolution microscopy techniques that it supports.