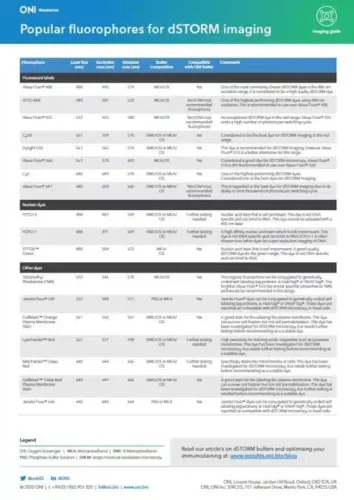
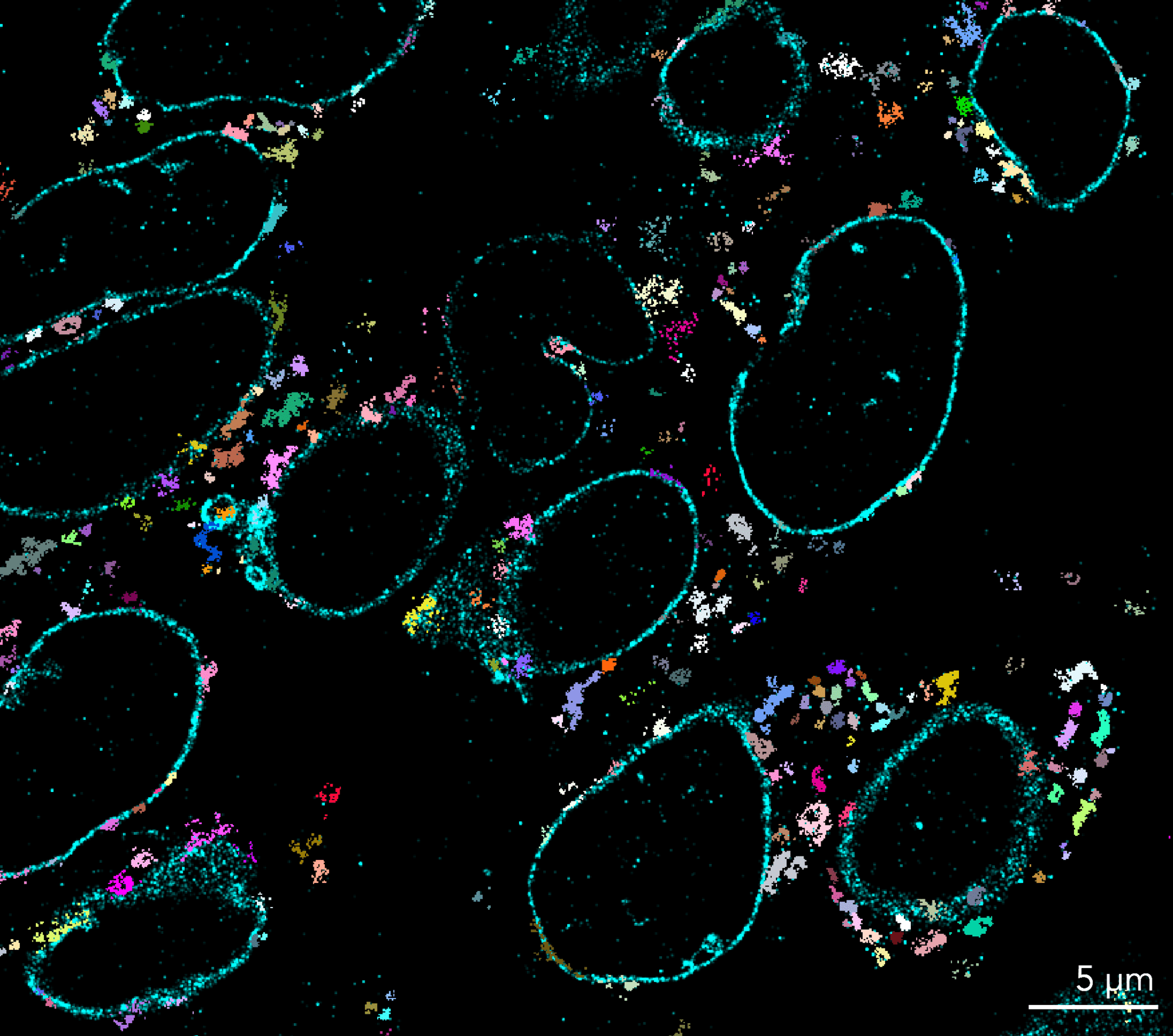
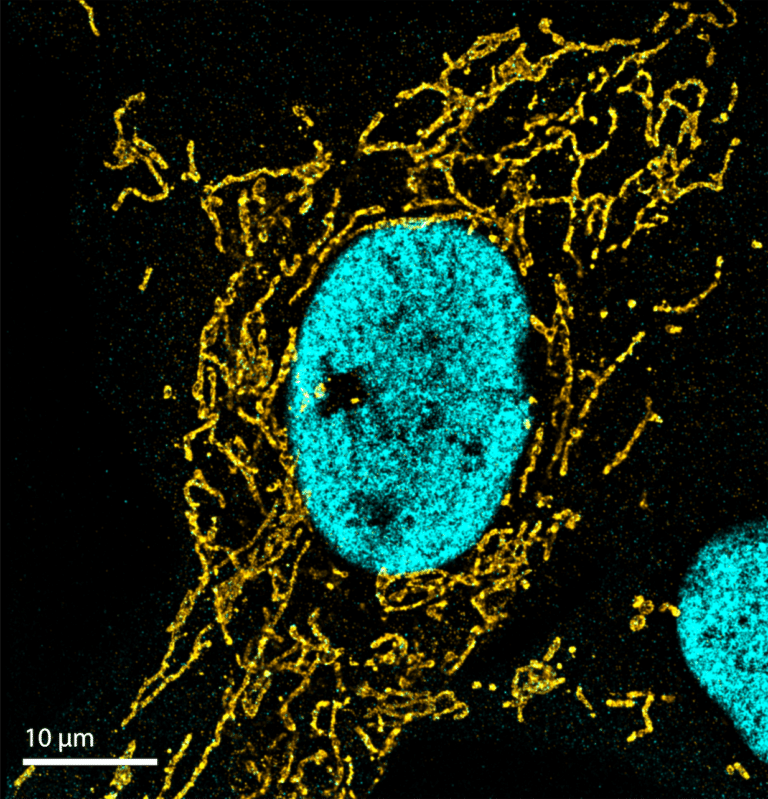
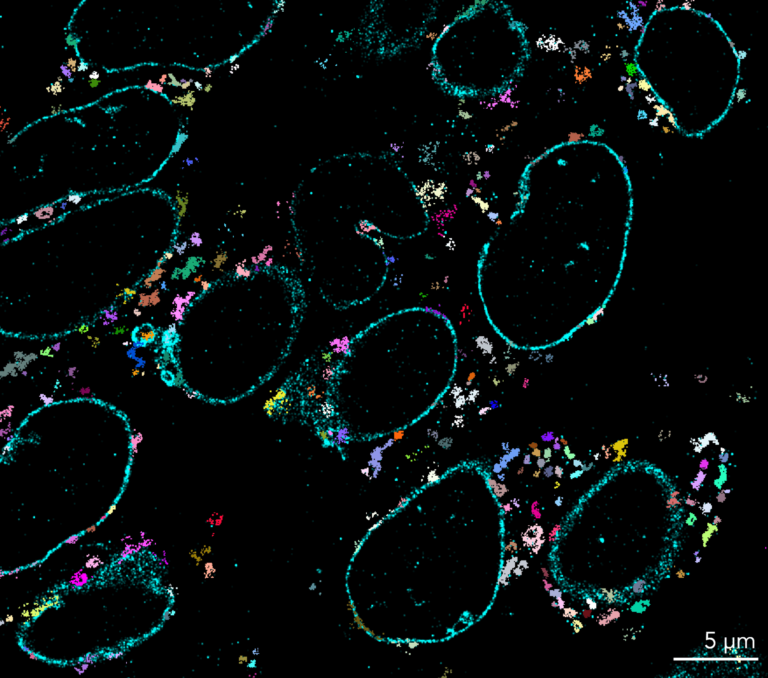
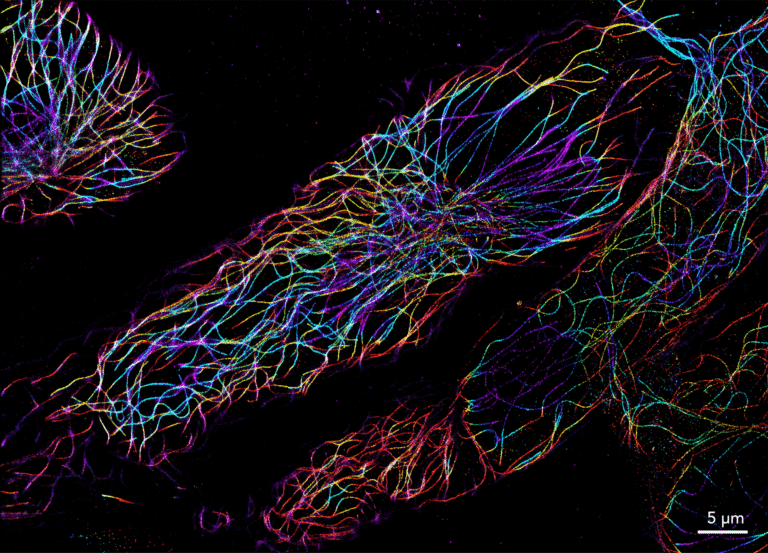
Since its invention in 2006, STORM microscopy has become the most widely used super-resolution microscopy technique for single-molecule imaging. STORM stands for “Stochastic Optical Reconstruction Microscopy” and it relies on the stochastic activation of individual fluorophores with photoactivatable properties. STORM is compatible with fixed cells and fixed particles and requires specific antibodies. It can generate images of molecular structures resolved to under 20 nm.
ONIs latest White Paper – Fast, high-throughput LNP sizing and characterization using an automated high-precision workflow read here>