Introducing: ONI’s Next-Generation End-to-End Solution for EV Research Learn More
Molecular Mechanisms and Interactions
Challenge
Understanding molecular interactions
Molecular interactions means the dynamic interaction between two or more labeled molecules. In cells, advanced microscopy techniques, and single-molecule FRET (smFRET) in particular, are used to understand the relationship between molecules through their spatial organization. For example, interest is growing in the underlying mechanisms of membrane contact sites (MCS) in cellular organelles.
However, often the most important aspect of an interaction is the rate of binding, the dissociation constant, the dynamics and dwell times. This quantitative information is especially useful for understanding the effect of inhibitors and small molecules or solution conditions on the behavior of a particular interaction, such as the activity of an enzyme induced by substrate binding.
Solution with the Nanoimager
Super-resolution microscopy applied to protein complexes
The Nanoimager boasts several features to support the study of protein complexes at the nanoscale level:
- Counting the number of subunits in a complex by intensity – the number of labeled proteins is equal to the sum of the intensity divided by that of a single fluorophore.
- Measuring the dynamics of assembling complexes, using single-particle tracking and/or smFRET.
- Counting subunits by the number of decremental steps as labeled proteins in a complex go dark due to photobleaching.
- Investigating the structure of protein complexes by super-resolution imaging, as has been rigorously demonstrated in the past with the nuclear pore complex and larger structures such as the cytoskeleton.
Famous examples of these methods applied to protein complexes include counting the subunits in the bacterial flagellar motor and in ion channels, as well as measuring the aggregation of amyloid beta associated with Alzheimer’s.

Case Study
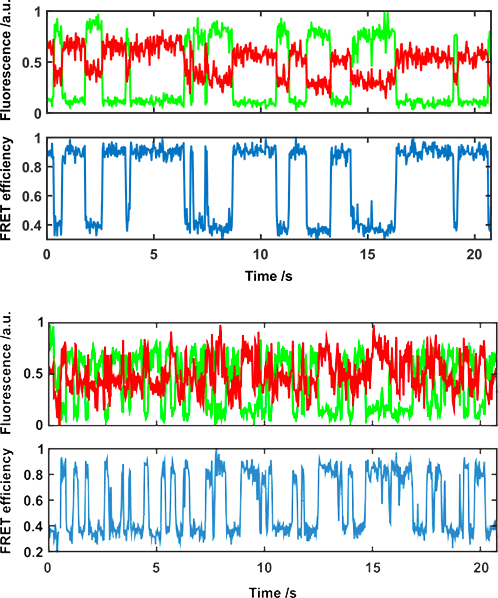
Single-molecule FRET
One approach used by the Nanoimager for extracting this information is single-molecule FRET. smFRET is a technique that involves labeling the sample with two fluorophores, which can be on the same or different molecular species, to measure a dynamic process (intra- and inter-molecular processes respectively). FRET acts as a real-time dynamic ruler on the 2-10 nm range, so it can detect two proteins binding and unbinding for example. It can also detect the conformational changes in a single enzyme, and how these transitions are affected by binding substrates.
The Nanoimager can measure these single-molecule interactions, and quantitate them, for various types of sample: molecules bound to a surface, in solution, on an artificial membrane or on a cell membrane close to a glass coverslip. Using TIRF, smFRET can be performed on live cell membranes with the Nanoimager.
In other methods, the Nanoimager can characterize molecular interactions using dynamic colocalization of two labeled species, monitoring their position over time.
Read more about the single-molecule FRET technique or read further super-resolution microscopy applications.